Primary Human Stellate Cells
Hepatic stellate cells have long been recognized as key players in liver homeostasis and pathogenesis.
The activation of stellate cells secondary to fatty liver disease and drug-induced liver injury is an accepted component of the progression of liver injury with fibrosis.
LifeNet Health LifeSciences offers primary hepatic stellate cells from both healthy and diseased liver tissues for use in research applications.
- Comprehensive donor medical and social history provided
- Histopathology report includes Non-Alcoholic Fatty Liver Disease (NAFLD) activity score (NAS) along with H&E and Trichrome stained images
- Donor-matched cells and tissue samples
- Cells from healthy and diseased tissue
- Detailed protocols and media recommendations provided
- Access to technical expertise and guidance from LifeSciences’ team of scientists
- Viability ≥70%
- Yield ≥0.5 X 106 cells/vial
- Immunocytochemistry (ICC) staining for GFAP, αSMA, and desmin
- Histopathology assessment of tissue of origin by board-certified pathologist; NAS* and fibrosis stage** provided
- Offered at Passage 0 and Passage 1
- Vials of cryopreserved cells shipped and stored at ≤-135°C
*NAFLD (Non-Alcoholic Fatty Liver Disease) score (NAS) was assigned according to the standards of the NASH CRN Scoring System (Hepatology 41: 1313-1321, 2005).
**Inflammation and fibrosis were assessed using standard Batts-Ludwig scoring methodology (Scale, 0-4, American Journal of Surgical Pathology 19: 1409-1417, 1995).
Human Stellate Cells are suitable for a range of applications, including
- Target identification
- Lead candidate screening
- Preclinical screening and testing
- Liver disease modeling, including Non-Alcoholic Steatohepatitis (NASH)
- Organotypic culture models – Co-cultures, 3D models
- Cell-cell signaling
- Mechanistic studies of Drug-Induced Liver Injury (DILI)
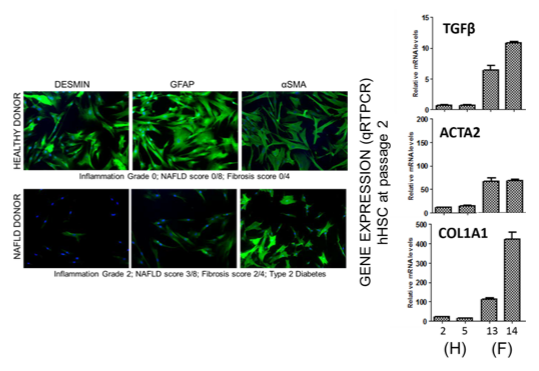
Characteristics of stellate cells derived from healthy- vs diseased-origin tissues
Each LifeSciences’ donor tissue is assessed and scored by a board-certified pathologist
with respect to steatosis, inflammation, fibrosis, and degree of Non-Alcoholic Fatty Liver Disease (NAFLD). Disease-origin tissues often display a greater degree of steatosis or fibrosis. Stellate cells isolated from disease-origin tissues retain some features associated with a disease-activated phenotype. Both morphologic and phenotypic differences can be detected. In general, expression of GFAP is diminished and SMA is increased in disease-origin cells. This suggests that at least some aspects of the disease phenotype are retained in the in vitro culture system.
Comprehensive Portfolio of Liver Cells
200+
Lots
0-6
NAS Scores
0-4
Fibrosis Stages
